You are here: vision-research.eu » Vision Research » The Young Researchers View » The Young Researcher's View: Anna Louise Vlasits (Q2-2022)
The Research of Anna Louise Vlasits
 |
In mammals, before visual information is transmitted to the brain, an intricate dance of visual processing takes place in the retinal circuit. Each of the cell classes in the retina—photoreceptor, horizontal, bipolar, amacrine, and ganglion cells—has multiple cell types, with the diversity of types especially large in the inner retina, where amacrine cells and ganglion cells each have over 30 types. These cell types are responsible for constraining and highlighting the visual information that mammals can use for vision. Despite great leaps in our understanding about the functions of individual cell types in the retina, many cell types’ roles in visual processing and behavior remain unknown [1]. My research focuses on two questions: what are the roles of these diverse neurons in visual processing and behavior? And how does genetic diversity between individuals, especially in the context of developmental disorders, alter visual processing?
I first encountered sensation as an student in Darcy Kelley’s lab at Columbia University, where I studied frog auditory behavior and brain anatomy. After graduating, I worked as a technician with David Raible and Edwin Rubel at the University of Washington in Seattle studying sensory hair cells in the zebrafish lateral line sensory system. These experiences cemented my interest in studying how neural circuits transform sensory signals from the environment to be useful for an animal.
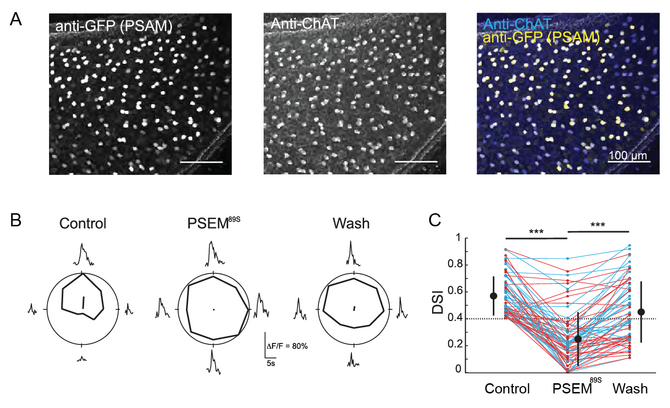
I began my focus on the retina during my dissertation research at University of California, Berkeley, in Marla Feller’s lab. There, I studied inhibitory amacrine cells in the retina called starburst amacrine cells, which are important for the retina to be able to sense the direction of motion. In a collaboration with John Flannery’s lab at UC Berkeley, we performed some of the first “pharmacogenetics” experiments in the retina to reversibly silence a single cell type [2]. We silenced starburst amacrine cells, and showed that their downstream synaptic partners, the direction selective ganglion cells, lose their motion direction tuning, confirming the starburst amacrine cells’ important role in this circuit.
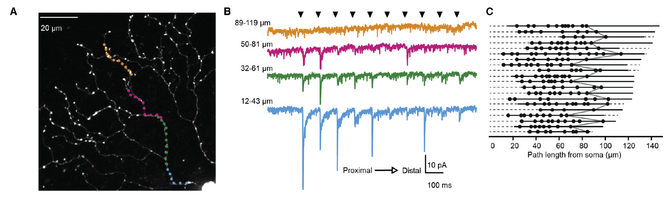
In a second study in collaboration with David DiGregorio’s lab at Institut Pasteur, I dived further into the function of starburst amacrine cells, which are non-traditional neurons because they lack axons. These cells have gorgeous radiating dendrites, where they are thought to compute motion information in each motion direction. How exactly this computation is performed was not totally understood, but it was thought that integrating the excitatory and/or inhibitory inputs would be critical. I used a technique called glutamate uncaging to map out the locations of the excitatory synapses onto these dendrites and found that the inputs are restricted to specific regions of the dendrites [3]. Using these results, I demonstrated how anatomical wiring and biophysical properties of starburst amacrine cells contribute to their ability to compute motion information.
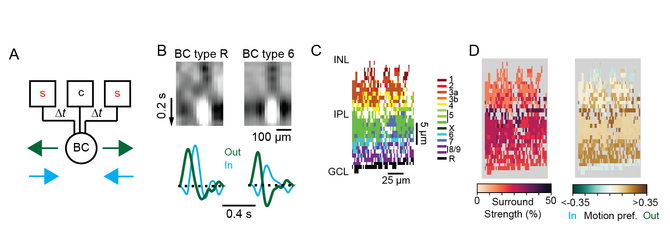
After zooming in to the level of individual synapses on a single cell in my PhD work, I decided to zoom out for my first postdoctoral position with Thomas Euler at University of Tuebingen. In collaboration with Philipp Berens’ group, I evaluated the functional properties of large datasets of bipolar cells’ and amacrine cells’ responses to visual stimuli using advanced computational methods and imaging techniques. By performing two-photon imaging of fluorescent sensors of glutamate, calcium and a novel voltage sensor [4], I uncovered previously unknown properties of retinal interneurons. In particular, I studied how diversity in the center-surround receptive fields of different bipolar cell types contribute to their responses to motion stimuli, and found that some bipolar cell types respond in a special way to motion stimuli and are radially direction selective (Fig. 3) [5]. In addition, through an ongoing PhD mentorship and collaboration with Philipp Berens and Katrin Franke’s groups, we are exploring how populations of amacrine cells, many of whose functions are still unknown, process chromatic information
Now, in Tiffany Schmidt’s lab at Northwestern University, I have zoomed out even more to study the link between retinal function and behavior. Because I am interested in how circuits operate in diverse genetic contexts, I am studying the retina in mouse models of neurodevelopmental disorders. I am using behavior techniques, cell-type specific genetic manipulation, electrophysiology, and in vivo calcium imaging to link visual behaviors to specific physiological and circuit-level differences in the early visual system mice carrying genetic mutations.
References
- A. Vlasits, T. Euler, K. Franke. Function first: classifying cell types and circuits of the retina. 2019. Current Opinion in Neurobiology. 56:8-15.
- A. Vlasits, R. Bos, R. Morrie, C. Fortuny, J. Flannery, M. Feller, M. Rivlin-Etzion. Visual stimulation switches the polarity of excitatory input to starburst amacrine cells. 2014. Neuron. 83:1172-1184.
- A. Vlasits, R. Morrie, A. Tran-Van-Minh, A. Bleckert, C. Gainer, D. DiGregorio, M. Feller. A role for synaptic input distribution in a dendritic computation of motion direction in the retina. 2016. Neuron. 89(6):1317–1330.
- C. Behrens, S. Zadav, M. Korympidou, Y. Zhang, S. Haverkamp, S. Irsen, A. Schaedler, A. Vlasits, K. Franke, K. Dedek, R. Smith, T. Euler, P. Berens, T. Schubert. Retinal horizontal cells use different synaptic sites for global feedforward and local feedback signaling. 2022. Current Biology. 32, 1–14.
- S. Strauss*, M. Korympidou*, Y. Ran, K. Franke, T. Schubert, T. Baden, P. Berens, T. Euler°, A. Vlasits°. Center-surround interactions underlie bipolar cell motion sensitivity in the mouse retina. °co-senior author. Preprint available: www.biorxiv.org/content/10.1101/2021.05.31.446404v2

Anna Louise Vlasits
Schmidt Lab
Northwestern University
Evanston, IL 60208
USA
Website: http://www.schmidtlab-northwestern.com
E-mail: anna.vlasits[at]northwestern.edu