You are here: vision-research.eu » Vision Research » The Young Researchers View » Oliya S. Abdullaeva (Q04-2016)
The Research of Oliya S. Abdullaeva
 |
My PhD project deals with optoelectronic solid-liquid interfaces and their potential to function as artificial photoreceptors. The prosthetic treatment of visual impairment has focused so far on the development of retinal implants based on inorganic electrode arrays and photodiodes. Alpha IMS (Retina Implant AG, Germany) and ARGUS II (Second Sight, US), which were approved for clinical trials, show promising results.1 Nevertheless there is a strong demand for alternative materials, as for instance good biocompatibility and effective electrical signaling are challenging issues.2,3 Especially due to their high biocompatibility, organic semiconductors are being considered as potential material for retinal prosthesis. 2,3
Our studies focus on squaraine dyes, in particular 2,4-bis[4-(N,N-diisobutylamino)-2,6-dihydroxyphenyl]squaraine (SQIB) (Figure 1), a small molecular model organic semiconductor blended with PC60BM ([6,6]-phenyl-C61-butyric acid methyl ester).
Squaraine dyes have a characteristic aromatic four-membrered ring system and due to their zwitterionic nature they are environmentally stable.4 They can absorb within the deep red of the visible light spectrum and therefore are employed as a photosensitive layer in photovoltaic devices.4 Light absorption leads to an excitation of electrons, therefore SQIB acts as an electron donor. In contrast, PC60BM functions as an electron acceptor facilitating charge separation. We grow N2A cells, a model neuronal cell line, on the SQIB:PC60BM-based artificial photoreceptor (Figure 2).5
Although undifferentiated N2A cells cannot generate action potentials like more complex neurons, they express voltage-gated sodium and potassium ion channels in their cell membrane. By performing proof-of-principle investigations we want to find out if our model organic semiconductor is capable of depolarizing N2A cells and induce ionic currents through ion channels when the semiconductor is stimulated by light (Figure 3).5
Our research has a more fundamental character as it focuses on investigating the signaling pathway of the photostimulation rather than fabricating an implant. Several mechanisms at the organic semiconductor/electrolyte/cell interface can play a role here, among them photoelectrical stimulation by capacitive charging of the interface,2,3 photothermal stimulation6 or even photochemical reactions taking place between the organic semiconductor and the electrolyte.
As a member of the DFG research training group “Molecular Basis of Sensory Biology” at the University of Oldenburg I have the great opportunity to work on a multifaceted, interdisciplinary project under the supervision of Jun.-Prof. Dr. Manuela Schiek from the Energy and Semiconductor Research Laboratory (Institute of Physics) and apl. Prof. Dr. Karin Dedek from the Neurosensorics group (Institute for Biology and Environmental Sciences). I conduct experiments on a regular basis in both Energy and Semiconductor laboratories and electrophysiology labs. SQIB is synthesized by the PhD student Matthias Schulz in the group of Prof. Dr. Arne Lützen from the Kekulé Institute of Organic Chemistry and Biochemistry at the University of Bonn, our long-term collaboration partner. Additionally, the stability of the samples and their surface morphology is further tested using atomic force microscopy and optical spectroscopy by our collaboration partner Prof. Dr. Frank Balzer from the Mads Clausen Institute at the University of Southern Denmark, SØnderborg. The stability tests show that the SQIB:PC60BM blend film interfacing with Ringer’s solution under dark conditions is stable for a longer period of time, although the degradation process is accelerated by illumination.5
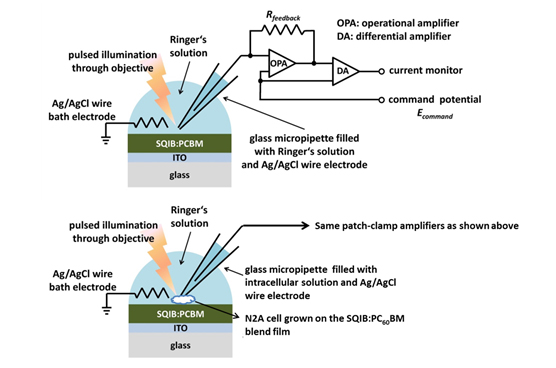
After the synthesis of SQIB, the devices are prepared by spincoating from solution on glass substrates coated with a transparent conductive oxide (ITO: indium tin oxide), and are subsequently annealed. We employ patch-clamp experiments (voltage- and current-clamp mode) to study the electrical response of the N2A cells during illumination of the SQIB:PC60BM based artificial photoreceptor and conduct transient photocurrent measurements on floating samples under physiological conditions in Ringer’s solution (Figure 3).5
Due to the high annealing temperature the SQIB:PC60BM blend film shows a textured crystalline morphology (Figure 2, middle) which is beneficial for cell growth, even without an adhesion layer (Figure 2, right).
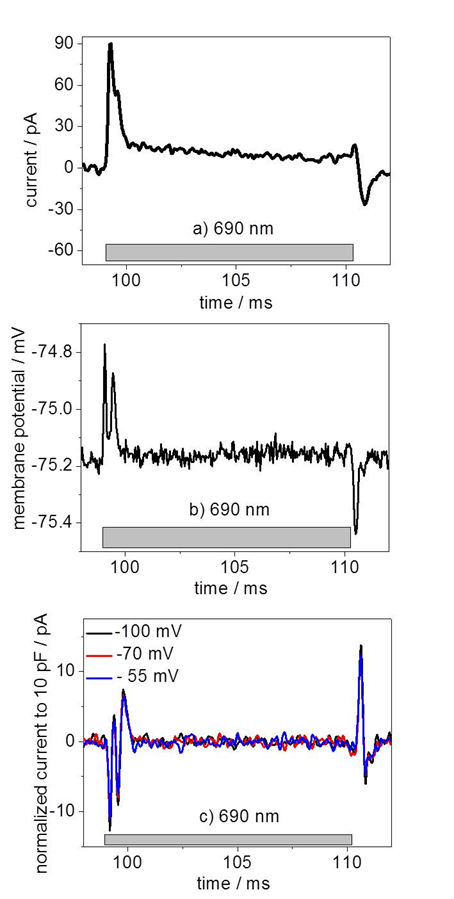
Transient photocurrent measurements in which the micropipette is held in close proximity to the SQIB:PC60BM blend film reveal fast capacitive displacement currents in the Ringer’s solution upon illumination for 10 ms from the electrolyte side, indicating an electrical coupling mechanism (Figure 4a)).5
By conducting the same photostimulation experiment on SQIB:PC60BM samples while simultaneously patch-clamping a N2A cell grown on the blend film we are able to induce a small change in the membrane potential of the N2A cells (Figure 4b)). This results in rapid capacitive transmembrane currents, which we record in voltage-clamp mode (Figure 4c)). The polarity of the fast capacitive displacement currents in the Ringer’s solution indicates an accumulation of negative ionic charge outside of the cell membrane which eventually depolarizes the cell.5
When the experiment is repeated with a SQIB:PC60BM blend film on bare glass without the ITO conductive layer, the capacitive displacement currents are absent and so is the depolarization of the cell which excludes any additional photothermal effect. Although we observed a slight change in the membrane potential, a depolarization below 1 mV is not sufficient to induce Na+-inward currents. We cannot trigger ionic current across the cell membrane yet but we achieved direct capacitive coupling between the N2A cells and the SQIB:PC60BM blend by photoelectrical stimulation without impairing the cell viability.5
The ineffectiveness of the photoeletrical stimulation so far demonstrates that we might need to change our device architecture in order to enhance the capacitive coupling effect, for example by introducing a dielectric insulating layer. Adjusting our experimental setups, by using higher light intensities or changing the illumination direction could also have an improving effect.
We might even need to consider the possibility that merely one mechanistic pathway is not sufficient, but that it is rather a combination of mechanisms, e.g., photoelectrical and photothermal, that eventually activate ion channels. Changing the experimental procedures by prolonging the illumination time for example may reveal additional photothermal effects.
References
- Hadjinicolaou, A. E. et al. Prosthetic vision: devices, patient outcomes and retinal research. Clin. Exp. Optom. 98, 395-410 (2015).
- Ghezzi, D. et al. A polymer optoelectronic interface restores light sensitivity in blind retinas. Nat. Photonics. 7, 400-406 (2013).
- Gautam, V. et al. A polymer optoelectronic interface provides visual cues to a blind retina. Adv. Mater. 26, 1751-1756 (2014).
- Hestand, N. J. et al. Confirmation of the Origins of Panchromatic Spectra in Squaraine Thin Films Targeted for Organic Photovoltaic Devices. J. Phys. Chem. C. 119, 18964-18974 (2015).
- Abdullaeva, O. S. et al. Photoelectrical Stimulation of Neuronal Cells by an Organic Semiconductor-Electrolyte Interface. Langmuir. 32, 8533-8542 (2016).
- Martino, N. et al. Photothermal cellular stimulation in functional bio-polymer interfaces. Sci. Rep. 5, 8911 (2015).
Oliya S. Abdullaeva
Optoelektronische Organik
Telefon: +49(0)441-798 3150
Email: oliya.abdullaeva[at]uni-oldenburg.de
Carl von Ossietzky Universität Oldenburg
Institut für Physik
Abteilung EHF
Carl-von-Ossietzky-Straße 9-11
D-26129 Oldenburg