You are here: vision-research.eu » Vision Research » The Young Researchers View » Slaven Erceg (Q03-2012)
The research work of Slaven Erceg
 |
Dr. Erceg has more than six years of postdoctoral experience dedicated to cell therapy involving human embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs), as a therapeutic tool for treatment of neurodegenerative diseases. He developed an improved protocol for differentiation of hESCs and iPSCs toward neural progenitors, oligodendrocytes and motoneurons, for the treatment of rat model of spinal cord injury (Erceg et al., 2008, Erceg et al., 2010a), and cerebellar neuronal progenitors for the treatment of animal cerebellar ataxia models (Erceg et al., 2010b, Erceg et al., 2011). Recently he has extended his interest to rare retinal diseases and is currently developing a study on the therapeutic potential of pluripotent cells in retinal degenerative disease involving gene correction
In total he produced 40 papers as main and co-author in high ranking journals like Stem Cells, Stem Cells and Development, Neuroscience, Hepathology etc
Currently he is holding the position of independent investigator contracted by Spanish Ministry of Health in Andalusian Molecular Biology and Regenerative Medicine Centre (CABIMER), Seville, Spain, and 2 research grants.
Development of patient specific iPS cells in order to model Leber Congenital Amaurosis disease
In progressive retinal degenerations (RD) such as LCA or RP, gene and small molecule therapy has been shown to be insufficient as neither of them can restore damaged or lost cells. Cell-based therapy has been shown to be a feasible approach for retinal degeneration as the transplanted cells can integrate into degenerating retina, form synaptic connections and improve visual function. This cell-based approach has been revolutionised by the development of stem cell technology which provides an unlimited source of pluripotent cells capable of differentiating into any cell type including retinal cells (retinal pigment epithelium (RPE) cells and photoreceptors). The generation of induced pluripotent cells (iPSCs) as patient-specific cell source offers an advantage in circumventing immune response and avoiding ethical issues related to the use of embryo-derived stem cells (ESCs). Both iPSCs and ESCs have been successfully differentiated to photoreceptor and RPE cells (Osakada 2009, Meyer J et al., 2009)
The wide genetic heterogeneity of RDs suggests a diversity of disease mechanisms which are largely unknown and the variability of individual responses to drugs cannot be predicted. iPSCs can be created for a variety of reported mutations in contrast to animal models which in addition bear interspecies differences. Modeling retinal pathogenesis in vitro using patients’ iPSC-derived retinal cells can elucidate disease mechanism and serve as a drug screening platform. The challenge is to select “normal” control lines with which to compare patient-derived lines, due to large subject variation. Ideally, pairs of isogenic lines, a disease-bearing one and another with corrected, or removed mutation should be compared.
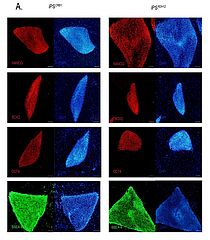
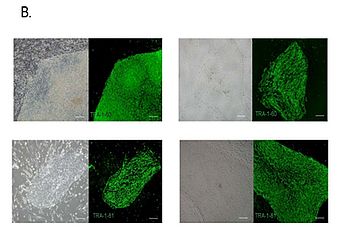
Here we have developed patients’ specific iPS cells by reprogramming two RD patients’ fibroblasts: one carrying compound heterozygous mutations in crumbs homologue 1 (CRB1) gene and another homozygous for a mutation in NADPH-dependent retinal reductase RDH12. We characterized the clones for pluripotency and ability to form all three germ layers. We aim to establish appropriate control lines with corrected mutations and subject the cell lines to differentiation toward retinal cells.
Slaven Erceg
Centro Andaluz de Biología Molecular y Medicina Regenerativa
Departamento de Terapia Celular y Medicina Regenerativa
Edif. CABIMER - Avda. Américo Vespucio s/n
Parque Científico y Tecnológico Cartuja 93
41092 Sevilla
Spain
Phone: +34 954 468 202
Fax: +34 954 461 664
E-mail:
slaven.erceg[at]cabimer.es
Website:
http://www.cabimer.es