You are here: vision-research.eu » Vision Research » The Young Researchers View » Jacqueline Butterworth (Q01-2010)
Major Research Projects
Project 1
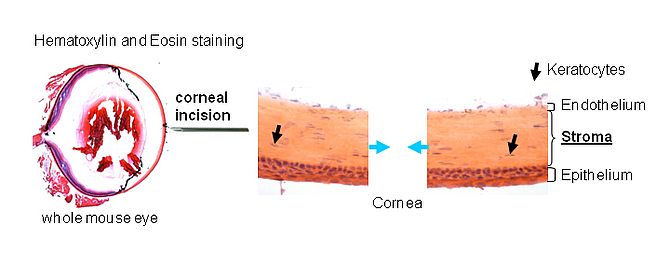
In vivo mouse cornea wound healing model: response of a stromal repair cell population
The outermost epithelial cell layer of the cornea is now more characterised for it’s continuous self-regeneration capacity and basal stem cell niche residing in the limbus. The corneal stroma consits of keratocytes embedded between collagenous lamellae responsible for the secretion of a unique matrix of glycoproteins and proteoglycans required for maintianing the integrity of the stroma. In response to acute injury the stromal keratocytes become mitotically active, adopt a fibroblast phenotype and migrate into the injured area to secrete new extracellular matrix. However, the regenerative capactity of the stroma in terms of a potential repair cell population remains unclear and less explored.
First reports of isolation of murine and bovine corneal stromal stem cells by sphere forming assay were made (2005) followed by localisation of an in vitro stromal subpopulation of ABCG2-postive cells in healthy human corneas, that coexpressed ABCG2 and PAX6, product of a homeobox gene not expressed in adult keratocytes. Afterwards, it was demonstrated in vitro that the human and rabbit stroma harbours CD133- and NESTIN-positive progenitor cells. Most recently, in vitro human corneal wounded tissue showed increased expression of stem cell markers OCT3/4, Telomerase and WNT-1 aswell as differentitation markers PAX6 and SOX2 in the corneal endothelium.
My work
The laboratory has developed an in vivo mouse corneal wound healing model upon full thickness incision of the central cornea. A selection of precursor markers has been used to determine the localisation and mobilisation of the potential repair niche in the stroma by means of immunohistochemical and RT-qPCR analysis of tissue from wounded and unwounded corneas at days 3, 7, 14, 21 and 28 after incision.
Project 2
High myopia candidate gene search
Myopia (short sightedness) is the most common eye disease and effects 25% of the world-wide population. High myopia is classified as a refractive error of ≤-6 D (Diopters) and an axial eye ball length of at least 26mm. It effects approxiamtely 3% of the European population and is the fourth cause of blindness due to secondary complications (retinal detachment, retinal haemorrhage, glaucoma...) (1).
Figure 2: The visiual symptom of a patient suffering from retinal detachment
The physiopathology of high myopia remains unclear, both genetic (20%) and environmental factors (80%) contribute to the complex trait of the disease. Previously our laboratory has shown a dominant mode of transmission of high myopia in French families, and then went on to localise linked chromosal regions on chromosme 7q36 and then 7p15.
Figure 3: Previously identified myopia and high myopia candidate genome regions
My Work
A GWAS (Genome-Wide Association Study) is underway on a newly established South-Western European Caucasian high myopic population in order to reconfirm the previous regions identified by our laboratory and identify novel genome hot spots using the high-throughput Affymetrix Array 6.0 SNP genotyping (Atlas Biolabs GmbH, Berlin).
The cases are compared to a control consortium from the Welcome Trust (Sanger Institute) so population stratification issues are being taken into account.
A first analysis of extreme high myopes (≤-12 D) is in progress to identifiy associated genomic regions that we aim to replicate in a second GWAS stage in a candidate region association study. A permanent national French and Spanish public hospital ophthalmology recruitment scheme is constantly extending the phenotype and DNA bank of high myopes and geogrpahically matched controls in Toulouse (France).
Bibliography
- Meng W, Butterworth J, Malecaze F, Calvas P (2009).
Axial length: an underestimated endophenotype of myopia.
J Med Hypotheses. in press - Fenwick A, Richardson RJ, Butterworth J, Barron MJ, Dixon MJ (2008).
Novel Mutations in GJA1 Cause Oculodentodigital syndrome;
J Dent Res. 87: 1021-1026
Jacqueline Butterworth
INSERM U563 FRANCE,
Marie Curie Research Training Network 'MY EUROPIA' MRTN-CT-2006-034021