You are here: vision-research.eu » Vision Research » Visionary of the Quarter » Thomas Euler (Q03-2014)
The research topics of Thomas Euler
 |
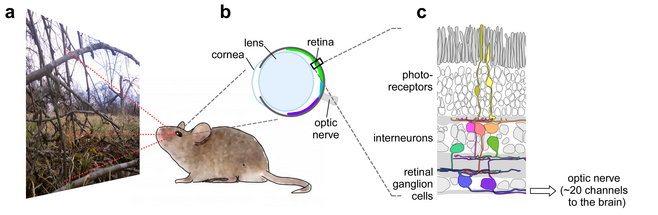
The visual system makes a tremendous effort to start analysing the incoming stream of images already in the periphery, that is, in the retina. Accordingly, the retina is a highly specialized and sophisticated image processor; it contains more than 70 types of neurons organized in at least 20 different visual feature-detecting microcircuits. Each microcircuit is represented by a different type of retinal ganglion cell (RGCs) processing the visual scene in parallel at every retinal location. This arrangement effectively generates an equal number of image representations, each encoding specific visual features, including simple properties such as contrast and "colour", but also more complex properties such as the trajectory of objects. Through the optic nerve, these "feature streams" are relayed to higher visual centres in the brain, where our view of the world is formed. The importance of retinal processing is highlighted by the fact that the optic nerve represents the main bottleneck in the visual system, therefore, the "decision" what visual features are relevant - i.e. for the survival of the animal - has to be made already in the retina.
My group aims at unravelling the function and structure of retinal microcircuits towards a better understanding of the underlying computational principles. Furthermore, we are interested in how these microcircuits change during retinal degeneration. Our cornerstone technique is two-photon microscopy, which we employ to image light-driven activity in retinal neurons - from the level of the individual synapse to the population level. To this end, we use synthetic and genetically-encoded fluorescent probes for intracellular ions such as calcium or chloride, as well as for synaptically released glutamate. These probes are "loaded" into the isolated retinal explant using electroporation or expressed in a cell type-selective manner in transgenic mouse lines using viral transfection. In combination with single-cell electrophysiology and anatomical techniques, we study the strategies that microcircuits throughout the retina use to extract visual information.
One of our current goals is to follow the visual information across the retinal layers along the vertical excitatory pathways. In particular, we study how the same set of visual stimuli is represented at the level of the photoreceptor and bipolar cell synaptic terminals, and how the information is integrated in the RGC dendrites, leading to the specific functional responses that are recorded in the RGC somata. We want to learn how visual features are extracted at different stages of this principal processing chain in the mouse retina.
Visual-feature extraction starts already at the first synapse
A distinctive property of the mouse retina is that the two spectral types of cone photoreceptors are not homogenously distributed across the retinal surface, but are arranged in a gradient: the "upper" (dorsal) half of the retina contains mainly green-sensitive cones and blue-sensitive cones are a minority, whereas the "lower" (ventral) half contains almost exclusively blue-cones (Fig. 1). Such a photoreceptor arrangement is not uncommon; it does occur in a number of vertebrate and invertebrate species, including rabbits, hyenas, guppies, dragonflies and some bees. The mouse offers an opportunity to study the functional consequences of this puzzling retinal property: the division into a more green- and a more blue-sensitive retinal part.
Using a combination of visual ecology, optical recordings, anatomy and information theory we recently showed that this division of the mouse retina has little to do with colour vision, but instead provides mice with an improved detection of achromatic contrast (Fig. 2). Measuring the cone output using two-photon calcium imaging in the isolated retina, we found that the responses of blue-cones in the ventral retina markedly differ from those of green-cones in the dorsal retina (Fig. 2a): the great majority of blue-cones showed higher gains (that is, they responded stronger to the same intensity light) and they responded best to dark contrasts (that is, decreases in light-level). In contrast, green-cone responses were much more balanced in their response to bright and dark contrasts. Because the lens of the eye inverts the image of the outside world (Fig. 1b), the blue-cones in the ventral part of the retina "look" at the sky. But it is not the match in colour (blue-cone vs. blue sky) that matters here, but the fact that statistically, dark contrasts are more common in the sky domain than bright contrasts: objects (e.g. branches, but also aerial predators) are typically dark against the bright backdrop of the sky. That this intuition captures the reality pretty well can be confirmed by measurements of contrast statistics in natural images (Fig. 2b-d).
Our data suggest that the mouse retina, with its differential cone distribution, is optimised for the different contrast statistics of ground and sky regions already at the cone photoreceptor level. In other words, mice appear to have put their different cone types to clever use: Different types of cones may be very helpful for efficiently detecting, for instance, an approaching aerial predator, which should appear as a dark silhouette in the mouse's visual world. Accordingly, dichromacy in some species may provide for more than mere "colour vision" - but in addition provide for the optimal sampling of achromatic contrasts in their natural environment and, thus, may offer an evolutionary advantage.
“Fingerprinting” the retina’s output
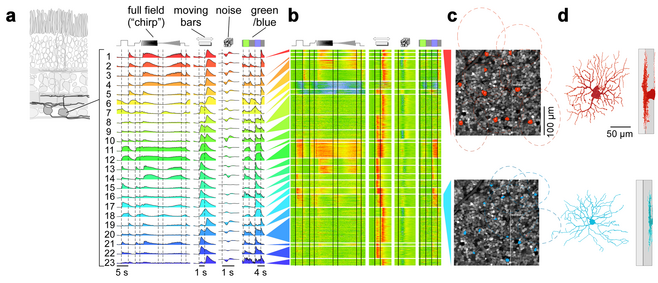
An important question pursued by many labs is what set of visual features does the retina encode. Our approach to this question is to simultaneously record from all RGC types at one retinal location using two-photon imaging (Fig. 3) to obtain a complete sample of the information sent to the brain and to understand how the representation of spatio-temporal information in a local image patch is distributed across the set of RGC types.
To this end, we have developed a battery of visual stimuli towards a physiological characterization of all cells in the ganglion cell layer in an approx. 300 by 300 µm patch of retina. Currently, our mouse database contains more than 10,000 cells from more than 30 retinas. Out of those, the best responding approx. 7,000 cells were used as the input to a Mixture of Gaussians Clustering model (Fig. 3a,b).
We obtained around 30 functional clusters, which roughly separate into 20 RGC clusters and 10 displaced amacrine cell clusters, as verified using immunostaining for GAD, the enzyme that marks GABAergic amacrine cells. These numbers match well the number of RGC and displaced amacrine cell types expected in mouse retina. The RGC types found include many known cell types (e.g. OFF and ON alpha, W3, ON-OFF direction selective; see e.g. Fig. 3c,d top), as verified using genetic labels, single-cell electrophysiology and other data available (e.g. soma size and retinal tiling). In addition, they include new functional types, including a contrast-suppressed type (Fig. 3c,d bottom) and an OFF orientation selective RGC. Our These results suggest that a “fingerprint” for each RGC in the mouse retina and, therefore, a functional description of the feature streams send from the retina to higher visual centres in the brain is within reach.
Recent publications
- Baden T, Euler T (2013) Early Vision: Where (Some of) the Magic Happens, Curr Biol 23(24):R1096-R1098.
- Baden T#, Schubert T#, Chang L, Wei T, Zaichuk M, Wissinger B, Euler T. (2013) A Tale of Two Retinal Domains: Near Optimal Sampling of Achromatic Contrasts in Natural Scenes Through Asymmetric Photoreceptor Distribution. Neuron 80(5):1206-1217, doi:10.1016/j.neuron.2013.09.030. # authors contributed equally.
- Baden T., Euler T., Weckström M., Lagnado L. (2013) Spikes and ribbon synapses in early vision. TiNS dx.doi.org/10.1016/j.tins.2013.04.006
- Puller C, Ivanova E, Euler T, Haverkamp S, Schubert T. (2013) OFF bipolar cells express distinct types of dendritic glutamate receptors in the mouse retina. Neurosci 243:136-48.
- Chang L, Breuninger T, Euler T. (2013) Novel chromatic coding from cone-type unselective circuits in the mouse retina. Neuron, 77(3):559–571.
- Baden T, Behrens P, Bethge M, Euler T. (2013) Spikes in Mammalian Bipolar Cells Support Temporal Layering of the Inner Retina. Curr Biol. 23(1):48-52.
- Wei T, Schubert T, Paquet-Durand F, Tanimoto N, Chang L, Koeppen K, Ott T, Griesbeck O, Seeliger MW, Euler T*, Wissinger B. (2012) Light-driven calcium signals in mouse cone photoreceptors. J Neurosci. 32(20):6981-94.
- Auferkorte ON, Baden T, Kaushalya SK, Zabouri N, Rudolph U, Haverkamp S, Euler T. (2012) GABA(A) receptors containing the α2 subunit are critical for direction-selective inhibition in the retina. PLoS One 7(4):e35109.
- Tanimoto N, Sothilingam V, Euler T, Ruth P, Seeliger MW, Schubert T. (2012) BK channels mediate pathway-specific modulation of visual signals in the in vivo mouse retina. J Neurosci. 32(14):4861-6.
- Borst A, Euler T. (2011) Seeing things in motion: models, circuits, and mechanisms. Neuron 71(6):974-94.
- Breuninger T, Puller C, Haverkamp S, Euler T. (2011) Chromatic bipolar cell pathways in the mouse retina. J Neurosci. 31(17):6504-17.
- Briggman KL, Euler T. (2011) Bulk electroporation and population calcium imaging in the adult mammalian retina. J Neurophysiol. 105(5):2601-9.
- Schubert T, Euler T. (2010) Retinal processing: global players like it local. Curr Biol. 20(11):R486-8.
- Margolis DJ, Gartland AJ, Euler T, Detwiler PB. (2010) Dendritic calcium signaling in ON and OFF mouse retinal ganglion cells. J Neurosci. 30(21):7127-38.
- Dedek K, Breuninger T, de Sevilla Müller LP, Maxeiner S, Schultz K, Janssen-Bienhold U, Willecke K, Euler T, Weiler R. (2009) A novel type of interplexiform amacrine cell in the mouse retina. Eur J Neurosci. 30(2):217-28.
- Schlichtenbrede FC, Mittmann W, Rensch F, Vom Hagen F, Jonas JB, Euler T. (2009) Toxicity assessment of intravitreal triamcinolone and bevacizumab in a retinal explant mouse model using two-photon microscopy. Invest Ophthalmol Vis Sci. 50(12):5880-7.
- Euler T, Hausselt SE, Margolis DJ, Breuninger T, Castell X, Detwiler PB, Denk W. (2009) Eyecup scope--optical recordings of light stimulus-evoked fluorescence signals in the retina. Pflugers Arch. 457(6):1393-414.
Dr. Thomas Euler
Professor for Ophthalmic Research
Email:
thomas.euler[at]cin.uni-tuebingen.de
Phone: +49 7071 29-85028
Fax: +49 7071 29-25011
Websites:
www.eye-tuebingen.de/eulerlab
www.cin.uni-tuebingen.de/research/euler
Contact
CIN - Centre for Integrative Neuroscience
Institute for Ophthalmic Research
University of Tübingen
Otfried-Müller-Str. 25
D-72076 Tübingen